|
Open hearth furnaces are one of a number of kinds of furnace where excess carbon and other impurities
are burnt out of pig iron to produce steel. Since steel is difficult to manufacture due to its high
melting point, normal fuels and furnaces were insufficient and the open hearth furnace was developed
to overcome this difficulty. Most open hearth furnaces were closed by the early 1990s, not least
because of their fuel inefficiency, being replaced by basic oxygen furnace or electric arc furnace.
Steel-making processes
Essentially the production of steel from pig iron by any process consists of burning out the excess
carbon and other impurities present in the iron. One difficulty in the manufacture of steel is its high
melting point, about 1,370ˇĆC (about 2,500ˇĆF), which prevents the use of ordinary fuels and furnaces.
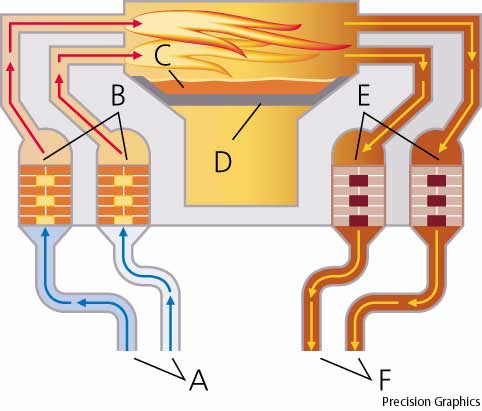
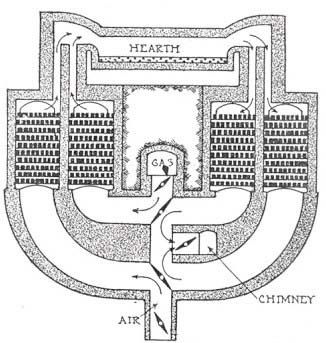
To overcome this difficulty the open-hearth furnace was developed; this furnace can be operated at a high
temperature by regenerative preheating of the fuel gas and air used for combustion in the furnace.
In regenerative preheating, the exhaust gases from the furnace are drawn through one of a series of
chambers containing a mass of brickwork and give up most of their heat to the bricks. Then the flow
through the furnace is reversed and the fuel and air pass through the heated chambers and are warmed
by the bricks. Through this method open-hearth furnaces can reach temperatures as high as
1,650ˇĆC (approximately 3,000ˇĆF).
The furnace itself consists typically of a flat, rectangular brick hearth about 6 m by 10 m (about
20 ft by 33 ft), which is roofed over at a height of about 2.5 m (about 8 ft). In front of the hearth
a series of doors opens out onto a working floor in front of the hearth. The entire hearth and working
floor are one story above ground level, and the space under the hearth is taken up by the heat-regenerating
chambers of the furnace. A furnace of this size produces about 100 metric tons of steel every 11 hr.
The furnace is charged with a mixture of pig iron (either molten or cold), scrap steel, and iron ore that
provides additional oxygen. Limestone is added for flux and fluorspar to make the slag more fluid.
The proportions of the charge vary within wide limits, but a typical charge might consist of 56,750 kg
(125,000 lb) of scrap steel, 11,350 kg (25,000 lb) of cold pig iron, 45,400 kg (100,000 lb) of molten
pig iron, 11,800 kg (26,000 lb) of limestone, 900 kg (2,000 lb) of iron ore, and 230 kg (500 lb) of
fluorspar. After the furnace has been charged, the furnace is lighted and the flames play back and forth
over the hearth as their direction is reversed by the operator to provide heat regeneration.
Chemically the action of the open-hearth furnace consists of lowering the carbon content of the charge
by oxidization and of removing such impurities as silicon, phosphorus, manganese, and sulfur, which
combine with the limestone to form slag. These reactions take place while the metal in the furnace
is at melting heat, and the furnace is held between 1,540ˇĆ and 1,650ˇĆC (2,800ˇĆ and 3,000ˇĆF) for
many hours until the molten metal has the desired carbon content. Experienced open-hearth operators
can often judge the carbon content of the metal by its appearance, but the melt is usually tested
by withdrawing a small amount of metal from the furnace, cooling it, and subjecting it to physical
examination or chemical analysis. When the carbon content of the melt reaches the desired level,
the furnace is tapped through a hole at the rear. The molten steel then flows through a short trough
to a large ladle set below the furnace at ground level. From the ladle the steel is poured into
cast-iron molds that form ingots usually about 1.5 m (about 5 ft) long and 48 cm (19 in) square.
These ingots, the raw material for all forms of fabricated steel, weigh approximately 2.25 metric tons
in this size. Recently, methods have been put into practice for the continuous processing of steel
without first having to go through the process of casting ingots.
|